 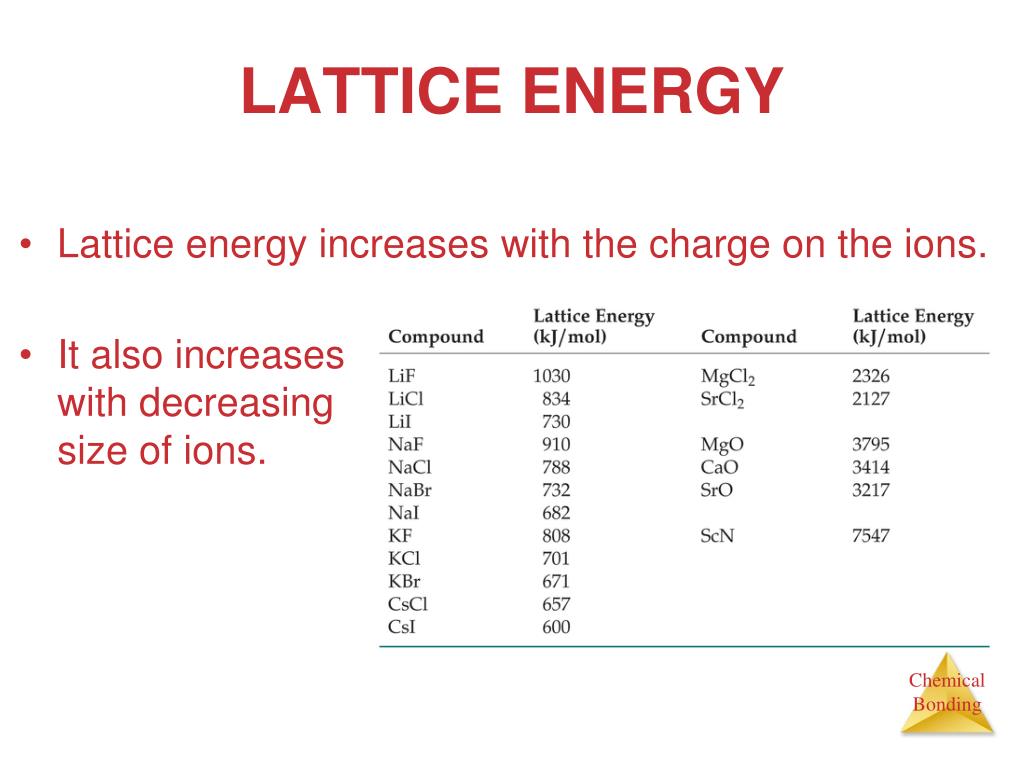
The Born–Landé equation above shows that the lattice energy of a compound depends principally on two factors: N A is the Avogadro constant M is the Madelung constant, relating to the geometry of the crystal z + is the charge number of the cation z − is the charge number of the anion e is the elementary charge, equal to 1.6022 ×10 −19 C ε 0 is the permittivity of free space, equal to 8.854 ×10 −12 C 2 J −1 m −1 r 0 is the nearest-neighbor distance between ions and n is the Born exponent (a number between 5 and 12, determined experimentally by measuring the compressibility of the solid, or derived theoretically). The relationship between the lattice energy and the lattice enthalpy at pressure P Following this convention, the lattice energy of NaCl would be +786 kJ/mol. as the energy required to convert the crystal into infinitely separated gaseous ions in vacuum, an endothermic process. Some chemistry textbooks as well as the widely used CRC Handbook of Chemistry and Physics define lattice energy with the opposite sign, i.e. In the case of NaCl, lattice energy is the energy change of the reaction The concept of lattice energy was originally applied to the formation of compounds with structures like rocksalt ( NaCl) and sphalerite ( ZnS) where the ions occupy high-symmetry crystal lattice sites. Lattice energy and lattice enthalpy Sodium chloride crystal lattice Since it generally cannot be measured directly, the lattice energy is usually deduced from experimental data via the Born–Haber cycle. The size of the lattice energy is connected to many other physical properties including solubility, hardness, and volatility. It is a measure of the cohesive forces that bind ionic solids. In chemistry, the lattice energy is the energy change upon formation of one mole of a crystalline ionic compound from its constituent ions, which are assumed to initially be in the gaseous state. When we are calculating lattice energy, we can use Born-Haber Cycle and when we have to compare the Lattice energy of the ionic solid, we have to keep both the factors in mind.Energy change upon the formation of one mole of ionic solid Note: Lattice energies are defined for ionic solids only. Hence, the compound having strongest lattice energy will be $LiF$. So, we have to move on the second factor, distance between the ions, as in the given cations $Na,K,Li$, $Li$ has the smallest size and in the anions $Cl,F$, $F$ has the smallest size. So, they don't differ by the charge on the ions factor. So, in the given options, cations and anions $Cl,F$, all the atoms have same charge, as the cations are of the same group (Group 1), +1 charge and all the anions of the same charge as they are of same group (Group 17: Halogens), -1 charge. Hence, smaller the size of the ions, greater the force of attraction, stronger the lattice stronger the energy required. Hence, the distance between the ions is inversely proportional to the electrostatic force of attraction. The distance between the ions decreases the electrostatic force of attraction. (b) Distance between the ions: As the lattices of the atoms are attached by the electrostatic forces of attraction. Hence, larger the charge, larger the force of attraction and stronger the lattice, stronger the lattice stronger the energy required. The electrostatic force of attraction is directly proportional to the magnitude of the charge held by the ions present in the ionic solid. 
(a) Charge on the ions: The lattices of the atoms are attached by the electrostatic forces of attraction. The lattice energy is expressed in the terms of Kilojoules per mole ( $KJmo$ ). It can be measured by using the Born-Haber Cycle. Lattice energy: The energy required to convert one mole of an ionic solid into gaseous ionic compound or the energy released by a crystalline solid when ions are combined to make a compound is known as Lattice energy. Hint: We have to find out what is lattice energy, how it is measured, how the lattice is attached, on which factors it depends upon, how the factors affect the lattice energy and then compare them with the given ionic solids.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
